News Snippets
The following news snippets were also published in the Wellington Botanical Society Newsletter
December 2018 News
From the President
Rodney John Lewington passed away on 27 September. Numerous BotSoccers attended his funeral on 3 October. Dr Patrick Brownsey represented the botanical community and spoke about Rodney’s extraordinary contributions to botany, in particular bryophytes, and to BotSoc. We offer our condolences to Darea Sherratt, and to the Lewington family. Please see the obituary in this newsletter.
We have had a wonderful programme of speakers recently:
17.9.2018. Susan Walker, Manaaki Whenua, Dunedin: the short tussock grasslands, shrublands, cushionfields and mat vegetation in the inland eastern South Island, and the threats to them.
15.10.2018. Bec Ramsay, WCC: draft plan for the Outer Green Belt. NIWA Science Fair winner, Thomas Fraser, Wadestown School: Maori rongoa / medicinal uses of native plants. Matt Biddick, PhD student: enlarged size of plants growing on islands, e.g., Tuhua (Mayor Island). Dr. Dave Burton: How to be sluggish, about the evolution of snails into leaf-vein slugs ideally shaped for reaching the moisture between long leaves such as on astelias and flaxes.
19.11.2018. Carol West: Plant of the month: horopito / Pseudowintera colorata. Lara Shepherd (Te Papa): Research on the genus Alseuosmia, toropapa. Several species of Alseuosmia resemble other plants, e.g., A. pusilla, which mimics horopito. Why do they resemble other plants? Stay tuned…
We thank all our speakers, and Sunita Singh who organises our meetings and field-trips programme.
Jon Terry, President
SUBMISSIONS MADE
Porirua Adventure Park
BotSoc lodged submissions with Porirua City Council and the Department of Conservation opposing the development and operation of an adventure park near Porirua on the grounds that it would be incompatible with the NZ Government’s ratification of the 2015 COP21 Paris Accord which requires NZ to slash greenhouse gas emissions. If the adventure park goes ahead, there would be a gondola with pylons, zipline, Base Station buildings, Top Station buildings, helicopter operations and new mountain bike trails. Permission has been sought for up to 55 helicopter movements per day. If the land managers give permission for the park, we asked for conditions to reduce the spread of weeds on the site, and for the proposed track in Rangituhi Colonial Knob Scenic Reserve to avoid the permanent vegetation plots established by DOC in the 1990s.
Wellington Conservation Management Strategy
The recently approved Wellington Conservation Management Strategy 2019–2029 will become available on 20 January 2019, the date the new CMS formally replaces the Wellington CMS 1996–2005. The Conservation Act 1987 requires the Director-General to review each CMS not later than 10 years after it was approved. The review process started in 2006/07, but since then has resembled a game of snakes and ladders. Local staff and the Wellington Conservation Board would start developing some ‘ladders’, but these would then be demolished or delayed by a ‘snake’ in the form of another restructuring, boundary changes, a new CMS template, and most recently, a change of government and a new Minister of Conservation. The statutory requirement didn’t seem to mean much; a succession of ministers kept approving extensions to the first CMS.
The draft of the latest CMS was released for public consultation in December 2016. BotSoc and 238 others lodged submissions on the draft in early 2017. (There is a summary of our key points in the May 2017 newsletter). After the public hearings on the submissions, DOC made ‘substantial changes’ before giving a revised draft to the Wellington Conservation Board which then worked with DOC to make further changes. The final changes came from the NZ Conservation Authority, and included a requirement for a policy and milestone for monitoring the effects of aircraft in one part of the conservancy (the Central Spine Place).
DOC and the Conservation Board have promised that this CMS has “more meaningful milestones to assist in the implementation and monitoring of the CMS”.
Proposed Regional Pest Management Plan (pRPMP)
We raised over 50 points in our submission on Greater Wellington’s pRPMP. (See the October 2018 newsletter for some of these). The analysis of submissions circulated by staff before the hearings included comments on nearly all of those points. Staff were quick to provide the Hearings Panel with species by species explanations of the reasons changes had been proposed to the status of pest species in the previous Regional Pest Management Strategy. In many cases, the change was to comply with the new regulatory categories, introduced to improve national consistency between all RPMPs. Other changes will result in resources being focused on Key Native Ecosystems (KNEs), instead of across wider areas. Some proposed changes, however, will reduce the potential impact of some weeds, for example:
• Spartina, upgraded from surveillance to eradication because it is “deemed possible to eradicate it, and important to do so”
• Chilean needle grass upgraded from surveillance to exclusion, because it is “not known to be present, but preventing establishment important”.
Officers did not support our request that deer, pigs and goats be reinstated in the site-led category, saying GW does not need rules in the RPMP to control these species in KNEs, or on private land if GW has the landowner’s permission. DOC can use provisions in the Wild Animal Control Act to regulate safari parks and deer-farming.
Staff ’s most disappointing response was the rejection of our suggestions for clearer wording and explanations because our wording was not “consistent with the national template for RPMPs”.
We may raise this issue with the relevant Ministers because we think conservation organisations and landowners should be able to understand how regional councils decide on pest control priorities. For example, the list of “harmful organisms” in Appendix 2 has no legal status under the pRPMP; it just shows species which GW may manage in KNEs.
Please see the article “Greater Wellington Regional Council report” elsewhere in this newsletter.
Parks Network Plan update
In August, staff presented GWRC’s environment committee with a 37-page summary of the public feedback obtained from GW’s Parks Network Plan Review Discussion Document. It was reassuring to find that pests / weeds / diseases were identified as the most significant issue for the regional parks over the next 10 years, and the public wants a more collaborative approach to support and enhance ecological corridors. Many responders place a high value on trail-based recreation opportunities (walking, biking, horse-riding and dog-walking). This was the first time I’d seen calls for more parking places for horse-floats, and more mounting-blocks near toilets. Turning the feedback into a Draft Plan will be a challenge given the diversity of issues and views. Consultation on the draft Parks Network Plan is scheduled for early 2019.
Regional Biodiversity Framework Workshops
GW held five workshops recently as part of testing interest in developing a region-wide biodiversity framework that would result in better collaboration between the many organisations, groups and individuals in the Wellington region who work to protect and enhance indigenous biodiversity. This initiative follows similar projects in Taranaki and Hawke’s Bay where many organisations have worked out how to achieve a shared vision, goals and desired outcomes.
A representative of the Hawke’s Bay project offered a memorable piece of advice:
“If you want to go fast, go alone. If you want to go far, go together”.
Bev Abbott Submissions Coordinator
A P (Tony) Druce’s Trip Book
Alison Druce has found the two missing pages from the Trip Book – Botanical Field Trips throughout NZ from 1934 to 1994. If you purchased a copy of the book, we hope that you will soon be able to access the pages on BotSoc’s web site, then print them and have them bound into your copy of Tony’s landmark document. The trips covered are nos. 243 to 252. We will advise you if and when this is possible.
We thank Alison Druce for searching for and finding the pages among Tony and Helen Druce’s papers, and Barbara Clark, BotSoc secretary for sixteen years, who arranged for the print runs of the book done several years ago, and for continuing her involvement in the matter.
The committee
Otari-Wilton’s Bush Report
The gardens are looking beautiful now, with lots of spring growth and flowers. You may have noticed that we have recently changed two areas in the gardens. After being donated numerous pallets of volcanic rock from the natural heritage exhibition at Te Papa, we have created a small garden underneath the large ngaio tree near the Cockayne lookout. We’ll be adding some more plants to this garden from our nursery which have been collected from Tongariro National Park and the volcanic plateau. One of the areas in the Alpine Garden has also been renovated with soil replaced and new plants added. We will soon start to renovate other areas in the Alpine Garden, to show off an expanded collection of alpines.
We are seeing more school groups coming to Otari. Our educator, Anneke Mace, has re-established the Otari Bush Guides. The Bush Guides are students from Otari School who come here once a week to learn about our native flora. When other schools visit they are taken on a tour with our Bush Guides. It is a successful way to engage students and forms a great link for us with the school next door.
Two horticultural interns from Kew Gardens have just spent time with us. Eve and Qamar are both very interested in NZ’s flora. We took them for a trip to the Wainuiomata water catchment where we have been monitoring two small populations of kohurangi / Kirk’s tree daisy / Brachyglottis kirkii var. kirkii. We have fenced off a small number of plants in each population to protect them from ungulate browse. All plants previously had signs of browse. They are all very healthy, the protected plants showing no sign of further damage. It would have been good to see them coming into flower, but alas not during this visit.
I received an encouraging e-mail from Colin Ogle (Whanganui BotSoc) thanking us for the tupari-maunga / cutty grass / Gahnia xanthocarpa that we had grown for Gordon Park Reserve, Whanganui. A Whanganui BotSoc member collected the seed for me from the single plant that is left at the reserve. Colin says the plant would have been fairly common to the region but has been seriously depleted by habitat loss. It was great to see a photo of the forty plants in the ground not far from the parent.
Karin van der Walt, our Conservation Science Advisor, has been busy with ex situ conservation work now in full swing at Otari. The new Lab is very exciting and enables Karin, often training other garden staff, to work on projects such as pollination of Bartlett’s rata / Metrosideros bartlettii, cryopreservation of swamp maire / Syzygium maire, pollination of other threatened plants (including orchids), and studying seed characteristics and pollen conservation.
It is well worth your visiting Otari at the moment. The gardens look great and it is an unbelievably good flowering year so far ….
Finn Michalak, Otari Native Botanic Garden, 160 Wilton Rd, Wilton, Wellington. Ph 475 3245, 027 803 0045. finn.michalak (at) wcc.govt.nz
Obituary – Rodney J Lewington (1935 – 2018)
It is with a heavy heart that we acknowledge the passing of Rodney J Lewington on 27 September 2018. The Society’s thoughts are with his wife, Darea Sherratt, and his family. Rodney will be remembered for his generous personality, his positive spirit and his everlasting sense of humour. He was always a good person to have in a tight situation. Over the years he acquired an extensive knowledge of things botanical which he delighted himself in sharing with other BotSoccers, especially the younger generations.
Rodney joined the Wellington Botanical Society in 1961, the same year he arrived in New Zealand from England. Since then, he has acted as Secretary of the Society (1974– 1978), President (between 1980 and 1987), Treasurer (between 1989 and 2013) and two years as Auditor (2014– 2016). Rodney contributed to more than 15 publications, numerous botanical field-trips, plant lists and specimen collection to national bryophyte collections at the Te Papa and the Allan Herbaria. In the 1980s. He was known as botanist A. P. (Tony) Druce’s right-hand-man on summer field-trips, organising food and cooking utensils, carrying heavy equipment, always being very efficient, and showing a casual relaxed positive attitude despite the length of the trips, the number of people, the weather, and the hazards involved. Since then, Rodney has attended religiously the summer field trips, bringing his extensive knowledge of the New Zealand flora and sharing his personal resources with fellow botanists. He was particularly known for his timely morning wake-up call: “porridge” that brought all the late sleepers on time for breakfast.
Rodney developed a passion and keen eye for bryophytes and became one of New Zealand’s top experts in liverwort systematics, co-publishing one taxonomic paper and cowriting the unpublished guidebook Mosses, Liverworts, Hornworts and Lichens: a guide for beginners. Rodney’s “behind-the-scene” contributions have been substantially acknowledged in numerous publications, including New Zealand reference books on mosses and liverworts: The Mosses of New Zealand by Beever et al. 1992, and A Flora of the Liverworts and Hornworts of New Zealand, Volume 1 by Engel & Glenny 2008. His patience, dedication, rigour and sense for details provided the bryologist community with thorough descriptions of many bryophyte species which will continue to be used.
Rodney has promoted the learning of botany and more specifically of ferns and bryophytes by leading numerous walks and talks, mentoring young botanists, and leading bryophyte surveys during BioBlitz events. He has contributed substantially to the training of a new generation of bryologists who admire and respect him not only for his extensive knowledge of the flora, but his commitment, energy, and passion for botany. Rodney’s 57 years of committed membership of the Wellington Botanical Society has enriched the knowledge of the Society, its members, and the New Zealand botanical community considerably. He will be sorely missed.
Pascale Michel and Owen Spearpoint
Greater Wellington Regional Council report
We are updating our Regional Pest Management Plan. In October we held public hearings on the draft document. The hearings panel then asked our Biosecurity team to clarify the matters raised, and to seek more information on them. The plan is being revised and corrected.
Weed control work is well advanced in our parks, reserves and Key Native Ecosystems (KNEs). Our focus is mainly on boneseed, blackberry, climbing asparagus, and a first round of treatment of tradescantia where required.
Woolly nightshade and moth plant annual inspections are being done, and blue passionflower will be looked for (all these species are Total Control species under our existing Regional Pest Management Strategy). Sites must have no plants present for nine consecutive years before we consider the target species to have been eradicated from a given site. We will also be controlling Senegal tea in several locations on the Kapiti Coast before Christmas, with work on Manchurian wild rice and purple loosestrife to be done also.
We are monitoring green-thistle beetle under our Biocontrol programme, and are checking the status of white admiral butterfly release sites in the region – these are bio-control agents for Japanese honeysuckle. We hope that their large size will mean that they establish and disperse quite fast around the region.
We are testing the use of a small drone for surveying sites and mapping weed locations so that we can go straight to them, rather than having to sweep the entire site. We hope that this tool will help to make us more efficient in our work!
Katrina Merrifield, Biosecurity Officer (Plants) Greater Wellington Regional Council, Te Pane Matua Taiao, 1056 Fergusson Drive, PO Box 40847, Upper Hutt 5140. T: 04 830 4411, M: 021 417 739, www.gw.govt.nz
Please see a summary of BotSoc’s submission on the proposed Regional Pest Management Plan in the “Submissions made” section in this newsletter.
Urban forest restoration ecology
Kiri Joy Wallace, PhD Student, University of Waikato, Environmental Research Institute. kwallace (at) waikato.ac.nz.
Restoring forest structure and composition is an important component of urban land management, but we lack clear understanding of the mechanisms driving restoration success. For my PhD thesis I studied two indicators of restoration success in temperate rainforests: native tree regeneration and epiphyte colonisation. While some studies have reported long-term responses to forest restoration, my analysis goes further by identifying the biotic and abiotic conditions that constrain the colonization of late-successional species into restored urban forests.
Framed within general ecological theory and supported by a robust statistical analysis, the paper resulting from my research identifies the primary ecosystem properties that drive ecological restoration success. Most importantly, I found that tree regeneration and epiphyte colonisation in urban forests was most strongly controlled by non-native weeds and fluctuating microclimate and that a critical threshold is reached at approximately 20 years after the initial planting. At this 20 year mark the tree canopy closes sufficiently to create optimal conditions for tree seedlings and epiphytes to successfully grow.
I owe a debt of gratitude to the Wellington Botanical Society for their support of my research and resulting successful thesis completion and publication of a research article in Ecological Applications (Walker et al. 2017).
ABSTRACT
Restoring forest structure and composition is an important component of urban land management, but we lack clear understanding of the mechanisms driving restoration success. Here we studied two indicators of restoration success in temperate rainforests: native tree regeneration and epiphyte colonization. We hypothesized that ecosystem properties such as forest canopy openness, abundance of exotic herbaceous weeds, and the microclimate directly affect the density and diversity of native tree seedlings and epiphytes. Relationships between environmental conditions and the plant community were investigated in 27 restored urban forests spanning 3–70 years in age and in unrestored and remnant urban forests. We used structural equation modelling to determine the direct and indirect drivers of native tree regeneration and epiphyte colonization in the restored forests. Compared to remnant forest, unrestored forest had fewer native canopy tree species, significantly more light reaching the forest floor annually, and higher exotic weed cover. Additionally, epiphyte density was lower and native tree regeneration density was marginally lower in the unrestored forests. In restored forests, light availability was reduced to levels found in remnant forests within 20 years of restoration planting, followed shortly thereafter by declines in herbaceous exotic weeds and reduced fluctuation of relative humidity and soil temperatures. Contrary to expectations, canopy openness was only an indirect driver of tree regeneration and epiphyte colonization, but it directly regulated weed cover and microclimatic fluctuations, both of which directly drove the density and richness of regeneration and epiphyte colonization. Epiphyte density and diversity were also positively related to forest basal area, as large trees provide physical habitat for colonization. These results imply that ecosystem properties change predictably after initial restoration plantings, and that reaching critical thresholds in some ecosystem properties makes conditions suitable for the regeneration of late successional species, which is vital for restoration success and long-term ecosystem sustainability. Abiotic and biotic conditions that promote tree regeneration and epiphyte colonization will likely be present in forests with a basal area ≥27 m2/ha. We recommend that urban forest restoration plantings be designed to promote rapid canopy closure to reduce light availability, suppress herbaceous weeds, and stabilize the microclimate.
Please see the full publication for details about my research:
REFERENCE
Wallace, K.J.; Laughlin, D.C.; Clarkson, B.D. 2017: Exotic weeds and fluctuating microclimate can constrain native plant regeneration in urban forest restoration. Ecological Applications 27: 1268 – 1279. https://doi.org/10.1002/eap.1520.
Kiri Wallace received the Wellington Botanical Society Jubilee Award in 2016.
Eleanor Burton
Darwin’s barberry
Commentary by Kevin Hackwell
The topic arose when answering some questions after my 10-minute presentation on the role of forests in sequestering carbon at the “Wellington beyond Paris” symposium on 15 September. I made the case for encouraging the regeneration of native forests under a gorse nursery – particularly in the western hill country of Makara / Ohariu Valley, etc.
As keen as I am to see natives do their thing, there is nevertheless no question that regeneration of natives under gorse is a much more rapid ecological process than under manuka or kanuka. I did work on this process for a MSc back in the late 1970s. Gorse is also a brilliant carbon sequester (~450 tCO2e/ha over 30 years).
While everyone was pretty happy with this in respect to gorse, I got a question about Darwin’s barberry. I answered that question by taking a similar long-term ecologist’s view of the role that many weedy woody species play in successional processes. I commented that although it is not as helpful as gorse, it nevertheless will eventually be overtopped and replaced by natives, so that if you take the longer-term view you might be better off allocating scarce resources to other problems.
There is no question that Darwin’s barberry is a pain in the proverbial, and is an invasive weed in NZ. It is appropriate that it is listed on the National Pest Plant Accord, and that Landcare Research has released the Darwin’s barberry weevil to act as a biological control – they have also released similar weevils to control gorse.
You don’t find 200-year-old Darwin’s barberry ‘forests’ in South America – because it is an early stage successional plant that requires reasonable light conditions for germination. When asking his question at the symposium Chris Horne mentioned the work of Dr Kate McAlpine. Kate concluded that both intact NZ podocarp-broadleaf forest and beech-broadleaf forest is relatively resistant to invasion from Darwin’s barberry. However, forest edges and canopy gaps are likely to be susceptible to invasion (https://www.doc.govt.nz/Documents/science-and-technical/drds329entire.pdf).
In the light of my comments at the “Wellington beyond Paris” symposium, Angus Hulme-Moir phoned me to discuss the Forest & Bird Wellington Branch’s work on controlling Darwin’s barberry. I made it clear to him that I wasn’t saying don’t bother controlling the barberry–just as I wouldn’t say don’t manage gorse in some circumstances.
The key thing is to be really clear about why you want to do the control. If you have an area you are already managing – or where you are observing an active invasion that you have the ability to counter – then controlling Darwin’s barberry makes sense. The same would apply for gorse.
When talking about the thousands of hectares to the west of the city where regeneration is already well on its way – I think it would be a great waste of resources to try to deal with areas of Darwin’s barberry that have already been established. So – horses for courses.
Within the city’s inner green-belt there would be a variety of circumstances where Darwin’s barberry control could make sense. However, over the extensive outer marginal farmland areas I would argue for letting nature take its course. I hope this clarifies my views on this.
Kevin Hackwell, Chief Conservation Advisor, Royal Forest and Bird Protection Society of New Zealand Inc.
Richard Herbert responds
Richard Herbert, Friends of Tawa Bush Reserves, chair of Tawa Community Board, and member of the Wellington BotSoc committee.
The corollary is then that we probably ought not “let nature take its course” in areas where Darwin’s barberry is not already established, and not let it get established in the first place if we can. Tawa currently has no Darwin’s barberry and long may that last, and we will proactively remove any outliers we find.
The problem with most of the Wellington rural area is that there is little “Intact New Zealand podocarp-broadleaf forest and beech-broadleaf forest”, rather it is retired marginal pasture areas that are being colonised by Darwin’s barberry, and perhaps more successfully, or at least more quickly, than the indigenous colonising species. On some of the poorer soils and wind-swept ridge areas of Wellington will Darwin’s barberry ever be overtopped?
And perhaps we ought not be encouraging its establishment – I saw on Thursday a fine Darwin’s barberry specimen in a garden bed alongside Ian Galloway Park’s club-rooms near Otari-Wilton’s Bush, and more above Kaiwharawhara Stream’s bank within Otari-Wilton’s Bush regeneration areas which will never be shaded out because of the light-well caused by the stream.
Let’s hope that the Darwin’s barberry weevil is successful. I’m hoping for the tradescantia weevil also!
Kevin Hackwell responds to Richard Herbert’s comments
I agree that in many areas where Darwin’s barberry is not already established, it may be a sensible strategy to prevent its establishment. The same would apply to gorse in some circumstances. My original comments were made in the context of the ability of the scrubby farmland to the west of Wellington to become a significant carbon sink if we allow and encourage the regeneration that in many areas is already well advanced. The area has the potential to sequester about 300,000 t CO2e per year.
I’m certainly not encouraging the establishment of Darwin’s barberry – what I am encouraging is a careful consideration of when and where to do control:
As I said above:
The key thing is to be really clear about why you want to do the control. If you have an area you are already managing – or where you are observing an active invasion that you have the ability to counter – then controlling Darwin’s barberry makes sense. (the same would apply for gorse).
When talking about the thousands of hectares to the west of the city where regeneration is already well on its way – I think it would be a great waste of resources to try to deal to areas of Darwin’s barberry that have already been established.
I’ll also be very happy if the barberry weevil works well.
Kevin Hackwell
Bev Abbott comments
There will always be differences of opinion about what to get rid of, and what to live with until natural processes take their course. Here’s some information from a Landcare web site, and some more questions about Darwin’s barberry:
“Darwin’s barberry (Berberis darwinii) comes from Chile. It first became established in New Zealand in 1946 and is now common from the central North Island to Stewart Island.”
If Darwin’s barberry didn’t become established until 1946, perhaps that’s why we don’t find 200-yearold Darwin’s barberry forests in NZ? Do we have any information yet about the longevity of Darwin’s barberry - how long it takes to be overtopped, and what species are successful over-toppers?
The Landcare web-site is ( www.landcareresearch.co.nz/science/plants-animals-fungi/plants/weeds/biocontrol/approvals/completed/darwins-barberry.)
Bev Abbott is a member of the Wellington BotSoc committee
Frances Forsyth comments
It is overtopped by mahoe, karamu, mapou, tarata, kohuhu, hangehange, fivefinger, etc.
Frances Forsyth is a professional ecologist and a member of the Wellington BotSoc committee.
Puffball sightings sought
There is an unnamed puffball that I have found growing in Wellington on mahoe / Melicytus ramiflorus in association with a liverwort. It appears to be parasitic on the liverwort, but does not appear to have been recorded before. Jerry Cooper (Landcare Research) sequenced the DNA and concluded “It needs more work but the data hint at it being more closely related to the ancestor of all puffballs globally than any of the currently recognised genera …”.
Most are only a few millimetres in diameter and large, often egg-shaped, specimens are less than 10 mm high. They are slow to mature, taking months, and withered husks often persist for a year. To determine how widespread these are, I am keen to hear of any sightings from anywhere. If you have seen it or want more information, or would like to be shown known sites around Wright Hill, Karori, please contact me, Tony Wills, tnywlls (at) gmx.com.
Tony Wills
September 2018 News
From the President
Kia ora.
On 20 August it was my great pleasure to take on the role of Wellington Botanical Society president. We were also charmed with a wonderful lecture by Prof. Bruce Clarkson on the subject of plant hybrids.
Growing up near Eketahuna on the banks of the Mangatainoka River, I took an early botanical interest by growing kowhai and oak trees from seed. Later I completed an honours degree in plant ecology. Since then I have mostly worked for the Department of Conservation on native plants and exotic weeds. Lately, I’ve been fascinated with lichens and wetland plants.
Jon Terry, President
From the past-president
August sees the end of my term as Wellington Botanical Society president. The society has a presidency limit of two years, enabling new ideas to come in with each new president. I have thoroughly enjoyed my term, and will be moving to the position of WBS secretary. I would like to welcome our new president, Jon Terry, and our new committee member, Julia Stace. I look forward to working with you both.
A registration form for our Summer Camp is in this newsletter. Please book early to avoid disappointment, and to help Mick and me with our planning.
Lara Shepherd, Past President
SUBMISSIONS MADE
We were expecting draft long-term plans (LTPs) from Greater Wellington Regional Council (GWRC) and Wellington City Council (WCC) because LTPs have to be updated every three years. Two additional consultations by GWRC, however, came as a surprise: an early review of the Parks Network Plan 2011, and the statutory consultation on the proposed Regional Pest Management Plan 2019– 2039. Both consultations raised some challenging issues. Both had unreasonably short timelines.
WCC’S LONG-TERM PLAN
Predator control
We supported WCC’s proposal to increase funding for predator control, but couldn’t resist pointing out that Predator-Free NZ promotional material rarely mentions the wider ecological benefits of predator control, e.g., more native plants will germinate if there are fewer possums, rats and mice to eat their flowers and seeds.
Weed control
We urged WCC to allocate extra funding for weed control, pointing out that back in 2011, McGlone, M., and Walker, S., had warned that the most troubling likely consequences of climate change included the arrival of new weeds and the increased invasiveness of existing weeds.
Biodiversity governance
We encouraged WCC to review its biodiversity governance systems. WCC consults extensively when developing specific strategies and plans, e.g., Our Natural Capital 7 (2015), but it puts much less eff ort into integrating those plans and reporting back on what’s been achieved.
Council’s response
We’ve received the standard letter, and it promised a more detailed response.
TOWARDS GWRC’S PARKS NETWORK PLAN (PNP) 2019–2029
Back in 2011, we welcomed GWRC’s plan to introduce one consolidated management plan for all eight regional parks, instead of continuing to review the park management plans, one by one. The new approach, set out in the Parks Network Plan 2011, also allowed for some park-specific policies and plans.
In a separate initiative under GWRC’s Biodiversity Strategy 2011–21, GWRC identified areas with special biodiversity values on public and private land throughout the region, then introduced the Key Native Ecosystems (KNEs) Programme to protect those values. There are KNEs in all regional parks. Next came detailed KNE plans to describe the work to be done over a three-year term in each KNE. The plan for Te Marua Bush is in the KNE Plan for Kaitoke Regional Park 2014–2017.
The reasons for the early review of the 2011 plan are not clear, but many issues, opportunities and proposals are flagged in three informative “Food for Thought” papers released in June 2018; a Parks Plan Discussion Document (74 pages), External Influences on Parks (47 pages), and Farming in Regional Parks (73 pages).
The farming paper, (p.63), shows forestry options are also being investigated:
At Kaitoke, planting manuka would cost about $4,100/ha, while planting a mix of forest species would cost about $42,500/ha. The total capital cost of planting the 74 ha grazed at this park in manuka would therefore be approximately $320,000, planting a mix of forest species approximately $3.1 million. For new plantings to survive, a maintenance cost of about $1750/ha per year for weed management for five years would also be required.
Submission
Our submission included the following points:
• we were pleased to find climate change was recognised as a key consideration for biodiversity planning
• we suggested additional staffing would be needed to support and supervise the expected increases in the number of volunteers
• we asked about GW’s plans for managing priority Myrtacaeae, both populations and iconic trees
• we urged caution in applying the following philosophy (”It may be that, over time, weeds are gradually overtaken by native plants, and that the speed of this may be acceptable”).
More plans for some parks
Given the 2011 intention to reduce the number of plans and hence consultations, it was disturbing to read that another type of plan is proposed, Park Environment Plans, with a 30–50 year horizon, to guide decisions on grazing, land retirement and restoration.
Next step
Two months’ consultation on the draft PNP is scheduled for early 2019.
GWRC’S PROPOSED REGIONAL PEST MANAGEMENT PLAN 2019–2039
Just two days after we lodged our PNP submission, GWRC asked for submissions on its 108-page proposed Regional Pest Management Plan (RPMP) 2019–2039, which was supported by a 200-page cost/benefit analysis. The timeline was just 28 days.
Understanding the proposed RPMP was hard work. We’ve urged GW to make the final RPMP easier for more people to understand.
• The purpose statement is: “to outline a framework for managing or eradicating specified organisms efficiently and effectively”. There was no foreword or executive summary to explain what this means.
• The significance of the word “specified” in the purpose statement became clear on page 21 where we learned that just 15 weeds are specified in the proposed RPMP. Three weeds are to be excluded. Five are to be eradicated. One is subject to progressive containment, and three to sustained control. These categories/ programmes relate to the Invasion Curve. Three other weeds will be controlled at sites in Lower Hutt.
• Appendix 2 (p. 94) contains a list of another 100 weeds called “Other Harmful Organisms”, e.g. Darwin’s barberry, pampas, and didymo. GWRC may control these weeds in KNEs and on some council reserves. It may monitor them, but the rules and systems in the RPMP won’t apply.
• The term ‘unwanted organisms’ is not the same as ‘harmful organisms’. It has a statutory meaning in the context of the RPMP, but is not defined in the Glossary.
Other themes in our submission
• We are opposed to removing feral deer and feral pigs from the RPMP; and treating them as ‘Other Harmful Organisms’.
• We think GWRC was too quick to dismiss climate change as a pest management issue.
• There was no mention of diseases such as myrtle rust.
• There is a list of seven advocacy and education functions but no objectives or targets
Overall
The proposed RPMP reads more like a plan for an organisation (GWRC) than a plan for a geographic region (Wellington). Given the multiplicity of organisations with pest management plans and responsibilities in the Wellington region, we think it’s time those organisations developed an integrated pest management plan for the region.
Next steps
We’ve indicated we may suggest changes to the lists of weeds before the hearings. We also want to examine the performance data in GWRC’s annual RPMS Operation Plan Reports.
GWRC STARTS REVIEW OF BIODIVERSITY STRATEGY 2011–2021
Just before we lodged the RPMP submission, GWRC sent out invitations to workshops entitled “Designing a shared way forward for our region’s biodiversity”. These workshops, in August and September, provide an opportunity for individuals and groups to contribute to the development of a region-wide biodiversity framework.
GWRC’S DRAFT LONG-TERM PLAN
See Chris Horne’s report about BotSoc’s submission on GWRC’s LTP in the May newsletter.
Bev Abbott, Submissions Coordinator
GWRC Pest Plants – review of the past few months
Winter is always a quieter time for pest-plant control activities, with lower ground temperatures and inactive growth periods of target species meaning it isn’t a good time to do control work. We have been reviewing the work done in the region’s network of Key Native Ecosystem areas last season, and planning what needs doing next season. This is often a continuation of the same work programmes, with perhaps differing priorities. We are also taking on some wetland sites under our Wetland Programme. These run for a three-year term, so the sites will change on a steady basis.
As you will know, the proposed Regional Pest Management Plan is being reviewed – we have been analysing the submissions we received and considering the suggestions requests.
In addition to this, we have made a submission in support of the proposal to introduce the gall mite Aceria vitalbae as a biological-control agent to assist with the management of the weed old man’s beard (Clematis vitalba). An effective biocontrol agent for old man’s beard (OMB) will help to slow the spread and impact of this highly invasive and difficult to control species. OMB’s rapid growth rate, widespread distribution across the Wellington Region, and highly invasive nature, means conventional methods of control are often unsuccessful and uneconomic, leaving biocontrol as the only effective longterm management option. GWRC is a contributor to and participant in the National Biological Control Collective (NBCC), and supports the establishment of biocontrol species. A successful biocontrol agent for OMB would ease the reliance on manual removal of the plant and the use of herbicides. Through the NBCC, GWRC is involved in a growing number of biocontrol projects and believes it is an essential part of the future of pest plant management in NZ.
Our Field Team has been working mainly in Queen Elizabeth Park (boneseed and rhamnus / evergreen buckthorn) and in East Harbour Regional Park (climbing asparagus control, most recently in the Butterfly Creek area). They have also done boneseed control in other parts of the region
Katrina Merrifield, Biosecurity Officer (Plants), Greater Wellington Regional Council Te Pane Matua Taiao, www.gw.govt.nz
2017/18 committee
At the 79th Annual General Meeting, held on 20 August 2018, the following were elected:
| Position | Name | Phone |
| President | Jon Terry | 021 168 1176 |
| Vice-presidents | Owen Spearpoint | 562 8780 |
| | Sunita Singh | 387 9955 |
| Secretary | Lara Shepherd | 384 7147 |
| Treasurer | Lea Robertson | 473 8211 |
| Auditor | Jane Humble | 971 6970 |
| Committee: | Eleanor Burton | 479 0497 |
| | Frances Forsyth | 384 8891 |
| | Richard Herbert | 232 6828 |
| | Chris Horne | 475 7025 |
| | Julia Stace | 385 4606 |
| Submissions coordinator | Bev Abbott | 475 8468 |
| Bulletin editor | Eleanor Burton
esmeraldadoris92 (at) gmail.com | 479 0497 |
President’s Report to the 79th Annual General Meeting of the Wellington Botanical Society
Welcome to the 79th Wellington Botanical Society AGM. The last year has seen us botanise the Volcanic Plateau, the Wairarapa, and many local sites as well. Finding new sites for us to explore is always a challenge. The map below shows Wellington BotSoc trip locations for the last year (in black) and since 2010 in grey. We certainly get around!
This year, thanks to the generosity of the Wellington 4WD Club, we were able to botanise an area of Te Kopahou Reserve that we would otherwise not have been able to access. Hopefully such collaborations can continue in the future, as they allow us to explore new areas rather than revisiting the same sites. Access to private land also provides new areas to visit and we thank landowners for their generosity in letting us botanise their properties. If you know of any out-of-the-way sites with interesting botany, particularly those with friendly landowners, then have a chat to our trip organiser Sunita Singh. We are also always after new people to lead trips – if you would like to volunteer then talk to Sunita and we can pair you up with an experienced trip leader.
Membership 2017/2018
Membership over the year has again remained broadly stable. We have 113 Ordinary Members, 31 Country Members, 61 Group Members, 30 Life Members, and 5 Student Members. The total membership figure stands at 240, slightly down from last year, but above the figure for 2015/2016.
Trips
The summer camp was well attended with 34 participants making the trek to Taurewa Camp on the Volcanic Plateau. The central location of the camp enabled us to visit a wide variety of habitats from the alpine zone to lowland forest, all within a short drive of the camp. Highlights for me included the range of alpine flowers on Mt Ruapehu and the hidden gem of Ohinetonga Scenic Reserve at Owhango. Thanks to Chris Moore for co-leading the trip and Richard Herbert and Leon Perrie for logistical support. I greatly appreciated Bev Abbott’s pre-camp advice on the catering. Graeme Jane and Mike Wilcox provided species lists and Chris Horne organised a collecting permit.
There were twelve other fieldtrips within the wider Wellington region last year. The most popular trips were to the Wainuiomata catchment (30 attendees) and Moa Point seaweeds (29 attendees). Thanks to Sunita Singh for organising the programme of trips, the landowners and to those who led trips, especially people leading trips for the first time this year.
Meetings
Sunita organised an engaging and educational programme of talks this year and I would like to thank her, as well as all our speakers for the effort they put into preparing and delivering presentations. Topics ranged from seaweeds to threatened plant listing, from Maori archaeological sites to the impacts of the Kaikoura earthquake on the region’s flora. Alex Fergus’s talk on the plants of the Subantarctic Islands drew the biggest audience this year with 58 attendees; Leon Perrie’s talk was a close second with 57 people turning up to discover more about ferns.
Communications
Newsletter: Three issues of the newsletter were produced during the last year – September and December 2017, and May 2018. Thank you to Chris Horne and Jeremy Rolfe for preparing, formatting and producing the newsletter.
Bulletin: Eleanor Burton is close to finalising the next volume of the Bulletin.
Website: Thank you to Richard Herbert and Julia White for your fantastic work managing our website and dealing with enquiries, respectively.
Facebook page: the Wild Plants of Wellington Facebook page continues to draw new members with membership growing from 237 this time last year to 341. It continues to be a good way to advertise our trips and meetings and to reach a new audience, particularly a younger demographic. It also serves as a forum to share news about plants, obtain plant identifications and recommendations and to start botanical discussions. Thanks to Julia White and Leon Perrie for administering the site.
Submissions
Bev Abbott spends a considerable amount of time preparing and presenting submissions on behalf of the Wellington Botanical Society. Thank you Bev for your enthusiasm for this role and to the committee for their thoughtful discussions on submissions. Thanks also to Chris Horne who prepared the Wellington Botanical Society submission on Greater Wellington Regional Council’s Draft Long-Term Plan in Bev’s absence.
Our newsletters include reports on some of the themes raised in our submissions on the following draft strategies and plans:
Greater Wellington Regional Council
• Draft Long-Term Plan 2018–28
• Draft Parks Network Plan 2019–2029
• Proposed Pest Management Plan 2019–39.
Wellington City Council
• Draft Long-Term Plan 2018–28
• Preliminary consultation Te Kopahou Reserve Management Plan
Upper Hutt City Council
• Draft Open Space Strategy 2017.
Awards
Matt Biddick received our Graduate Student Grant, which will aid his study of the morphological differences between offshore island plants and their mainland relatives. Kat de Silva received the Jubilee Award. She is studying factors that constrain or promote urban reforestation in revegetation projects, and how these change over time. We awarded two prizes at the NIWA Science Fair: Katie Harford’s project examined native New Zealand plants vs bacteria, and Lucy Hegan and Sarah Scott looked at sea lettuce and the reduction of nitrogen in polluted water.
Thanks to all the Wellington Botanical Society members who provided plants and books for sale at our meetings, and to those who bought them as this contributes to these awards.
Committee
The committee met regularly over the past year, mostly at the Leonard Cockayne Centre at Otari-Wilton’s Bush, with this venue organised by Eleanor Burton. I would like to acknowledge the committee for the behind-the-scenes work they do to ensure the smooth running of the society.
Thank you Owen Spearpoint (Vice-President), Frances Forsyth (minute taker), Bev Abbott (submissions coordinator), Barbara Clark (secretary), Lea Robertson (treasurer), Sunita Singh (organiser of field trips and speakers), Eleanor Burton (editor, BotSoc Bulletin), Richard Herbert, web site maintenance, and Chris Horne (editor, BotsSoc newsletter). I would like to especially thank Barbara Clark, who steps down as secretary this year but who will leave big shoes to fill. Barbara has been secretary for 17 years and we will miss her organisation and experience in this role. In recognition of her contribution the committee has made her an honorary life member of the Wellington Botanical Society.
Other thanks and acknowledgements
In addition to the people acknowledged above, I would also like to thank the following:
• Barbara and Kevin Clark for hosting the annual committee BBQ at their house.
• Sunita Singh, Trudi Bruhlmann, Ian Goodwin, Jill Goodwin, Lea Robertson, and Chris Horne for mailing out the newsletter and Kaaren Mitcalfe for providing a venue.
• NZ Print for their work printing the three issues of the newsletter.
• Victoria University of Wellington for the use of Murphy Lecture Theatre 101 for holding our meetings.
Lara Shepherd, President
FIELD TRIPS
| Date | Location | Attendance |
| 2.9.2017 | Forest of Tane, Tawa | 19 |
| 7.10.2017 | Saline wetland, Wairarapa | 19 |
| 4.11.2017 | Moa Point seaweeds | 29 |
| 11.11 2017 | Te Marua Bush workbee | 16 |
| 4–11.1.2018 | Taurewa Camp, Volcanic Plateau | 34 |
| 3.2.2018 | Te Kopahou Reserve | 23 |
| 3.3.2018 | Boulder Hill, Belmont Regional Park | 23 |
| 7.4.2018 | Wainuiomata catchment | 30 |
| 5.5.2018 | ‘Tanah Burung’, South Karori Rd | 22 |
| 9.6.2018 | Paekawakawa Reserve & Oku St Reserve, Island Bay | 16 |
| 7.7.2018 | Manawa Karioi, Island Bay | 12 |
| 14.7.2018 | Te Marua Bush workbee | 14 |
| 4.8.2018 | DOC Covenant, Makara Farm | 22 |
| | TOTAL | 279 |
| | AVERAGE | 21 |
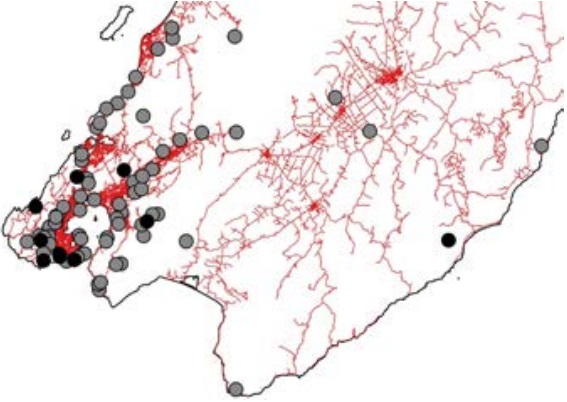 |
WBS field trip locations.
Black dots are sites visited in the last year.
Grey dots are sites visited since 2010. |
EVENING MEETINGS
| Date | Subject | Attendance |
| 17.7.2017 | C Moore; Stephen Hartley | 42 |
| 21.8.2017 | 1. Annual General Meeting
2. Druce Memorial Lecture: Rodney Lewington, Tony Silbery, Chris Horne | 35 |
| 18.9.2017 | Wendy Nelson | 47 |
| 16.10.2017 | 1. Nathaniel Walker;
2. Stacey Bryan | 35 |
| 20.11.2017 | Jill Rapson | 42 |
| 19.2.2018 | Matt Ryan | 42 |
| 19.3.2018 | Leon Perrie | 57 |
| 16.4.2018 | Alex Fergus | 58 |
| 21.5.2018 | Members’ Evening | 24 |
| 18.6.2018 | Jan Clayton-Greene | 44 |
| | TOTAL | 426 |
| | AVERAGE | 43 |
Annual Report from the Treasurer, Wellington Botanical Society for the year ending 30 June 2018
The accounts for the financial year ended 30 June 2018 show a surplus of $1,719 on the normal operations of the Society recorded in the General Account. Website invoices were not received, and although publication of the Bulletin has been delayed, provision is made for the following issue. An increase in postage expenditure reflects advance purchase of Kiwistamps and prepaid envelopes given a postal price increase from 1 July 2018.
Transfer of $960 each year from the General Account to the Victoria University Student Field Grant Account represents rent of the lecture room we use for meetings. The University allows the Society to use the room for our monthly meetings free of charge.
On the income side, subscriptions received have decreased by $360. Some memberships lapsed, and more members made advance payments late last year. Interest received increased. Currently we have an average interest return of 3.89% p.a. on invested funds up from 3.71% p.a. last year.
$2,600 was awarded from the Jubilee Award Fund, $600 was given as a Victoria University Student Field Grant, and $150 was awarded to a school student at the NIWA Wellington Science Technology Fair 2017. Jubilee Award Fund donations remained steady, and our sales effort improved markedly. Funds are in place to cover Bulletin 57 printing costs in the 2018/2019 financial year.
The full performance report has been filed with Charities Services, and will be loaded on their website https://www.charities.govt.nz/.
Lea Robertson, Hon. Treasurer
Seed germination, dormancy and longevity in the endangered shrub Muehlenbeckia astonii (Polygonaceae)
Debra M. Wotton, Moa’s Ark Research, Paraparaumu, Biological Sciences, University of Canterbury, Christchurch, Landcare Research, Lincoln, New Zealand
Abstract
Muehlenbeckia astonii Petrie (Polygonaceae) is a nationally endangered shrub undergoing widespread recruitment failure in the wild. Seed germination, dormancy and longevity were investigated to determine factors potentially constraining M. astonii regeneration and population persistence. Muehlenbeckia astonii seeds were collected from restoration plants sourced from Kaitorete Spit, Canterbury. Germination percentage of untreated control seeds in a growth room (20degC with 16 hours of light daily) was compared with that of seeds in two treatments: (1) chilled at 4degC for 2 weeks; and (2) chilled for 12 weeks. I also investigated seed longevity by comparing germination of unburied, 6-month old, cold-stratified M. astonii seeds with germination of seeds buried in the field and retrieved after 1–4 years. All seeds were sown in pots outdoors. In the growth room, 24% of untreated M. astonii seeds germinated, while cold-stratification for either 2 or 12 weeks increased germination significantly (to 76%). In the seed longevity experiment, 85.2% of unburied M. astonii seeds sown 6 months after collection germinated. Muehlenbeckia astonii seeds survived burial under the soil in the field for up to 4 years. However, germination declined rapidly to 27.6% after burial for 1 year, and only 2.8% of the seeds germinated after 4 years of burial. The high germination of cold-stratified seeds suggests that low seed viability is not limiting M. astonii regeneration at Kaitorete Spit. Muehlenbeckia astonii has the potential to form a soil seed bank, which may buffer small, isolated populations from local extinction in the short term (< 5 years).
Source: NZ Journal of Botany 2018 Vol.56, No.3 331-341.
Debra’s research was funded in part by a grant from BotSoc’s Jubilee Award Fund.
Otari-Wilton’s Bush report
Kia ora koutou.
I’m pleased to announce the appointment of Megan Ireland as gardener based here at Otari. Megan comes from a background of horticulture in England and Scotland. She has also worked in some NZ gardens, including Dunedin BG, for Annabel Langbein in Wanaka, and has worked twice previously at Otari. Megan replaces Nas Mulligan who moved to a horticultural position in Christchurch.
We have almost completed the establishment of the Lions Otari Plant Conservation Laboratory, 90% of the lab equipment has arrived, resource consent has been granted and building consent will follow soon. The laboratory will support plant conservation through research into seed storage behaviour, assessing seed viability, in-vitro propagation and establishing germination protocols.
The Lions Karori Club initiated funding for this project. I thank them for the funding they have made available and for their genuine interest and perseverance with this project. The laboratory project is a milestone for us. No other botanic garden in NZ has this type of facility. It will enable us to do work we have not been able to do before, increase our ability to partner with others and show that we mean business in ex-situ plant conservation.
The laboratory heralds the beginning of possible further changes to our nursery. We’ve recently embarked on a project with Victoria University School of Architecture to look at possible changes. Landscape architecture students have been developing concept designs for the nursery and surrounds. We would like to upgrade the nursery infrastructure and equipment, rearrange the nursery layout to improve efficiency and safety, and integrate the nursery into our visitor experience. The concept designs will be displayed at the Central Library, on the mezzanine floor near Clark’s Cafe from 14–23 September. The concepts will be subject to public voting, so if you are passing the Central Library, please pop in for a quick look and vote for your favourite.
With the creation of Te Tauihu, Te Reo Maori policy for Wellington City Council we now have a platform from which to incorporate Maori into our signs and interpretation. Over the coming months and years you will notice this beginning to show as we replace old signs and install new ones.
Otari Open Day is shaping up well. Our programme includes the usual plant sale (10 a.m.), Otari-Wilton’s Bush Trust BBQ and stalls. Capital Compost is joining us this year, along with a pop-up market in the Leonard Cockayne Centre featuring nature-inspired goods by local makers. In the Te Marae o Tane Information Centre the Open Lab will feature real lab equipment to investigate what creatures are out there on a microscopic level.
Hope to see you there.
Kind regards.
Rewi Elliot, Team Manager – Otari-Wilton’s Bush, rewi.elliot (at) wcc.govt.nz
Baring Head, East Harbour Regional Park
The 2018 planting season has almost finished. With one more planting day due we will have 2600 seedlings planted by volunteers, including a large group from the Garage Project brewery. As I reported in the last newsletter, these were mainly riparian plantings by the Wainuiomata River, with priority given to protecting and enhancing several inanga spawning sites. This was the first year that we obtained crowd funding through the Sustainable Business Network’s Million Metres programme. This has proved so successful that we’ve decided to increase the effort in 2019 to 5,000 plants, so we’re seeking funding from the public. Greater Wellington have contracted Remutaka Prison to propagate them and Conservation Volunteers will organise and oversee the plantings.
If you would like to contribute, then go to https://millionmetres.org.nz/donate/?project=2019-wainuiomata-river-restoration-at-baring-head. We’ve received a very generous offer from a private donor to match contributions up to $15,000, so your support will go a long way.
Colin Ryder, Treasurer, Friends of Baring Head
Obituary – Roger Michael Greenwood 1920–2017
 |
Michael Greenwood. |
Michael Greenwood, a life member of Wellington Botanical Society since 1953, died in Palmerston North on 14 August 2017. Until his retirement in 1980, he worked at Plant Chemistry Division and Applied Biochemistry Division, DSIR, where his work on isolating and typing Rhizobium bacterial strains, measuring their ability to nodulate their specific legume hosts was an important scientific development crucial to the productivity of NZ pastures.1
He also co-authored papers with another BotSoc luminary, Dr Ian Atkinson, notably seminal papers in the 1970s and 1980s suggesting the moa hypothesis for the unusually high frequency of occurrence of divaricating shrubs and juvenile trees in NZ.2
Born in New Plymouth, he initiated a career in biological science in Havelock North guided and inspired by respected amateur botanist and teacher, Norman Elder. He went on to contribute to the scientific eff ort of World War II, in work on improving the quality of dehydrated vegetables being supplied to troops in the Pacific, and also on the newly discovered hormone-type weed-killers and dangers of trace contamination.3 He built a house at Atawhai Road near Massey University and developed the now covenanted Greenwood’s Bush where Landcare Research employee John Innes did his Master’s thesis on rats.4 In his will, he bequeathed his Atawhai Road planting to the C.T. Keeble Memorial Forest Trust.
The Esler, Greenwood and Robertson family off spring were early, enthusiastic members of the Junior Naturalists’ Club, which he was instrumental in establishing. In 1984, he was involved with the “Save the Odlins Block Committee” which, through negotiation, saved a 2000-hectare area of native bush at Tokomaru from being logged. The area has since been returned to Tararua Forest Park. In 1993, he was awarded the Loder Cup for his outstanding contribution to the conservation of our unique flora. In 1997, he received a Civic Honour for community service, particularly to education and science, and in 2010, he was awarded Honorary Life Membership of the New Zealand Ecological Society for his scientific achievement, contribution to restoration ecology5 and to Keeble’s Bush, which is a 15.5-hectare protected bush reserve considered to be the finest remaining example of podocarp/broadleaf lowland forest in the Manawatu. Mr Greenwood was inaugural chair of the C.T. Keeble Memorial Forest Trust. He worked to provide for the security of the Bush, developed or overviewed its two associated restoration plantings, and continued throughout his lifetime to care for their flora and well-being.6
Lea and Hugh Robertson
1 Tribute to RM Greenwood, Trilepidea 166, September 2017, www.nzpcn.org.nz/publications/Trilepidea-166-170923.pdf
2 Ecological Society newsletter, 134, December 2010, pp 6–7 https://newzealandecology.org/sites/default/files/EcolNews_December_2010_134.pdf
3 Dr Jill Rapson ( pers comm )
4 Biology and ecology of the ship rat, Rattus rattus rattus (L.) in Manawatu (N.Z.) forests, Master of Science in Zoology, Massey University, https://mro.massey.ac.nz/handle/10179/6614
5 NZES Awards for 2010 – abridged nomination by Dr Jill Rapson https://newzealandecology.org/sites/default/files/EcolNews_December_2010_134.pdf
6 ‘Two areas of about five hectares adjacent to the Bush were also developed to greatly improve the ecological resilience of the original Bush remnant. Many small remnants of native vegetation persist today on agricultural land, and are assuming increasing importance as New Zealand's biodiversity declines and human impacts increase’ http://www.massey.ac.nz/massey/about-massey/subsidiaries-commercial-ventures/massey-agricultural-experiment-station/keebles-farm/keebles-farm.cfm
Queen’s Service Medal awards
We congratulate Sue Millar, Glennis Sheppard and Allan Sheppard who received QSMs in the Queen’s Birthday Honours for their work in conservation. All are members of Forest & Bird’s Upper Hutt Branch, and of BotSoc. Since 1992, Sue and Glennis have co-lead workbees in Te Marua Bush, usually accompanied by Allan. They have all also worked in Hulls Creek, Barton’s Bush and Domain Bush in Trentham Memorial Park, and Keith George Memorial Park, on weed and pest-animal control.
The committee
MEMBERS’ EVENING – 21 MAY 2018
Jill Goodwin
Jill read about nikau from NZ’s Native Trees (Dawson & Lucas. 2011, page 508), that “After the seedling has developed a few leaves, the growing point of the stem curves over and downwards to a depth of c. 15 cm. The deep growing point continues to be connected to the mostly emergent fronds, eventually growing upwards to emerge above the soil. Many large fronds are formed, but a trunk does not develop for 15 years or more, above and below ground. So in this way the base of the trunk ends up firmly embedded in the ground, like a post in a post hole.”
Sumitra Manga
Sumitra described her ideas for increasing the public’s awareness of BotSoc through possible changes to our web site.
 |
Lycopodium fastigiatum. |
 |
Ranunculus lyallii. |
Sarah Wilcox
Sarah showed slides taken in the South Island high country of Gastrodia sp., Aciphylla sp., Lycopodium volubile, Huperzia varia, Celmisia sessiliflora, Coprosma atropurpurea and Astelia nervosa, and on the beech-forest floor, a moss, Dicranoloma sp., and a lichen, Pseudocyphellaria sp.
Eleanor Burton
Eleanor showed some of the 550- 600 pottery tiles she has made for the path outside her back door. They feature images of native plants. She also showed some of her pen-and-ink drawings, e.g., Helichrysum dimorphum.
Chris Horne
Chris read from Canterbury Botanical Society Journal 48 2017, part of Miles Giller’s article: “Restoration planting – Are we saving or sabotaging our precious native remnants?”:
“Nature is far from random. Naturally occurring vegetation sorts itself out according to ecological patterns and processes. Various species occupy particular niches, some are generalists and others are quite specific in terms of where they will or won’t flourish. Different species have differing preferences or tolerances of soil types, aspect, rainfall, drainage, fertility, temperatures and shade, and juxtapose themselves in time and space accordingly. Canterbury has a broad range of habitat opportunities, stemming especially from its broad ranges of landforms and climates. Quite subtle site differences can have a strong bearing on what species prevail where. Plants often establish though a successional process, some species are tough pioneers capable of prospering under exposed conditions, others require a protective framework of associated species. Composition is rarely static, plant associations at any place can change with time, with additional species recruiting and others dropping out. Interdependence can occur at unseen levels, mycorrhizal and faunal associations can be pivotal in determining the health and survival of certain plant species.
Differences also occur at the genetic level, with plants at each site evolving local characteristics reflecting local selective pressures. For some species, like kowhai Sophora microphylla, this has led to quite distinct local forms. Each remnant provides a vignette-like representation of the former vegetation patterns at that particular site – at both species and genetic levels. Each is subtly unique, and if we are to truly respect and protect their ecological integrity then we need to avoid actions that might compromise them.
If we are to do any planting, the species selection, placement and timing needs to be very accurately managed. It can be argued that nonlocal native species or provenances are just as problematic as exotic weeds, as any such plant simply takes up space and resources that might otherwise be occupied by a representative local species. This can be especially problematic if the non-local species is fecund, as their progeny can occasionally become aggressive volunteers. At least non-local species can usually be identified and removed, however if they hybridise with local species or are otherwise difficult to distinguish then they can be very difficult to manage. The North Island lacebark Hoheria sexstylosa has caused major problems in several South Island sites, volunteering aggressively and hybridising with the local Hoheria angustifolia where present, all at the expense of representative local species.
The practice of restricting plant selection to local provenances goes some way to addressing the issue. However the presence of a species at one locality does not necessarily mean that it is suitable for a nearby locality, as the two sites may have quite different habitat characteristics. Further, the importance of selecting local provenances is inconsistently recognised and applied. Some nurseries, advisors and planters are keen advocates whilst others are ambivalent. The ideal provenances are not always available when wanted, and substitution with alternative species or provenances is not uncommon. There is often a temptation to purchase plants on price rather than provenance, as we can get greater numbers from cheaper lines, allowing us to achieve greater coverage."
May 2018 News
From the President
This year’s Wellington BotSoc summer camp 4–11 January was very well attended with thirty-four members making the trek to Taurewa Camp. The camp, situated between National Park township and Turangi, is Avondale College’s outdoor education centre. Accommodation is in ex-forestry huts sleeping two to six.
Apart from torrential rain on our first day of botanising, the weather was mostly co-operative. The camp’s central location enabled us to visit a wide variety of plant habitats from the alpine zone to lowland forest, many only a short drive away. Mike Wilcox, Auckland Botanical Society, provided excellent advice on day-trip options, which made our planning much easier.
Thanks to Graeme Jane and Mike Wilcox for providing species lists, and Richard Herbert for keeping track of transport costs. Bev Abbott’s pre-camp advice was a huge help to Lara’s organisation of breakfasts and lunches. Last, but certainly not least, a big thank you to Susan Brown and her two (adult) children, for their outstanding dinner catering and hosting. We highly recommend Taurewa Camp to any other botanical society, or other large group, wanting accommodation.
The 2019 summer camp will be based at Bannockburn, central Otago, where the vegetation will provide quite a contrast to this year’s camp. We are booked for 25 January to 1 February at Bannockburn, so please pencil it into your diaries if you plan to attend.
Lara Shepherd, President
SUBMISSIONS MADE
Taupo Swamp – background
In early 2016, BotSoc lodged a submission with Greater Wellington Regional Council (GWRC), asking that Taupo Swamp be treated as an Outstanding Wetland instead of as a Significant Wetland in GWRC’s Proposed Natural Resources Plan. BotSoc later advised GWRC that we no longer wish to speak in support of our submission, because we are satisfied with the assessment provided by Dr Philipa Crisp (GWRC), and the recommendation in the Section 32A report. More importantly, we do not have any new technical information to bring to the panel’s attention.
Evidence
Two reports provided to the hearings panel since we lodged our submission provided more thorough descriptions and assessments of Taupo Swamp’s biodiversity values.
• Queen Elizabeth II National Trust provided a report commissioned from Wildlands Consultants;
• Dr Philipa Crisp provided technical evidence on Wetlands and Biodiversity (see below). She sought advice from Shona Myers in preparing her evidence.
Section 42A Report
This vital report, prepared by Guest and Denton, recommends adding the Taupo Swamp Complex to Schedule A3, with a consequential change to delete it from Schedule F3 “Significant natural wetlands”.
Hearings
The Hearings for Hearing Stream 5 began on 9 April for three weeks, covering sections of the plan on Beds of lakes and rivers; Wetlands and biodiversity; Discharges to land.
Bev Abbott, Submissions Coordinator
If you would like to read the entire submission, including appended statements by Dr Philippa Crisp, please contact the editor.
Greater Wellington Regional Council’s Long-Term Plan 2018–2028
GWRC’s vision for freshwater quality and biodiversity: “The quality of the freshwater in our rivers, lakes and streams is maintained or improved, and our region has healthy plant, bird and wildlife habitats.”
Our society strongly supports this priority, and welcomes the fact that this is the first in the list of the council’s four priorities. We believe that healthy indigenous ecosystems – aquatic and terrestrial – are of fundamental importance to our region, and ultimately to New Zealand and the planet.
GWRC’s placing of environmental protection at the top of the council’s list of priorities is entirely consistent with our society’s Rules, in particular our Object 2(d).
Management of regional parks and forests
We are concerned at the adverse impacts of track building and road building on natural landforms, indigenous ecosystems and their supporting soils; we are also concerned at the adverse impacts of those activities on aquatic ecosystems.
Our East Harbour, Kaitoke and Belmont regional parks, Akatarawa Forest, Pakuratahi Forest, Battle Hill Farm Forest Park, Queen Elizabeth Park, and Whareroa Farm, have all been affected to some extent. Sometimes the building of tracks is done by GWRC, sometime by authorised community groups, and sometimes by unauthorised groups or individuals. Some tracks may be built for walkers and runners, others, usually wider, for ‘shared use’ by walkers, runners and mountain-bikers. Other tracks, in effect bush roads, may be built for off-road vehicles including 4WD vehicles, quads and trail bikes, and some are built for horses.
Recommendations
GWRC gives top priority to protecting indigenous plant communities, and their supporting soils and landforms on the lands it manages on behalf of the public, ahead of providing for recreational uses of those lands.
1. GWRC uses all means at its disposal, including the prosecution of offenders, to stop unauthorised construction of tracks and roads on any of the lands it manages on behalf of the public.
2. GWRC closes all unauthorised tracks and roads.
3. GWRC advertises in the print media and on its web site any proposals for the construction of new tracks and roads on the lands it manages.
Chris Horne, for the committee
Plague skink
This Australian reptile, Lamphropholis delicate, also known as rainbow skink, grows to 6–8 cm long. It is brown or grey-brown, often with a dark-brown stripe along either side of its body, with an iridescent sheen in a bright light. It has been found on Rakino Island, in the Hauraki Gulf. It serves as a warning that new invasive animals are arriving in NZ, small and mobile enough to get to our special places. Carefully check the contents of your pack before you land on any offshore island. Plague skinks and/or their eggs could easily be transported in potted plants. See article in Professionsl Skipper magazine by Amanda Peart, Auckland Council. See also www.treasureislands.co.nz.
Frances Forsyth
Percy Scenic Reserve news
We went on a seed-collecting trip 16–19 March, this time to Arthur’s Pass, John Van den Hoeven and me from Downers, and Jonathan Bussell from Hutt City Council.
We spent 16 March at Temple Basin, Arthur’s Pass. Like last year’s trip to Cobb Valley, there is a huge number of plant species in quite a small area. We managed to find seed from 38 species, including Celmisia armstrongii, Gentianella corymbifera and Ranunculus lyallii.
We spent 17 March on the screes of Foggy Peak, Porters Pass, where we collected ten species, plus about thirty species as cuttings, e.g., Azorella hydrocotyloides, Carmichaelia monroi and Kelleria dieffenbachia.
In two days we collected seeds of 49 species and cuttings of 41 species. With some cross over, we ended up with 76 species, with 48 being new to the collection.
We sowed all the seed over 10–11 April after processing them, and giving them a fortnight in the fridge. The first two batches started to germinate over 26-27 April, these being Epilobium melancaulon and Acaena glabra.
Also several of the cuttings have taken, and now have roots, including Carmichaelia monroi and Kelleria dieffenbachii. We have potted on some of the propagated plants from our 2017 Cobb Valley trip as they were becoming too big for their pots.
We have taken more cuttings of our Atriplex cinerea to add to our populations which we planted out last year, and are doing very well indeed.
Cliff Keilty, Percy Scenic Reserve
Worldwide Day of Botanical Art, 18 May 2018
‘Linking people with plants through botanical art.’
This project was proposed by the American Society of Botanical Artists to focus on plant diversity, to build connections with botanical artists worldwide, and to document some of the wild species of the world.
Juried exhibitions by each participating country to consist of original botanical artworks of the participating country’s native flora. Twenty-five countries are participating.
A digital collection of images from ALL exhibitions will be shown at each participating venue, alongside that country’s original artworks.
See www.botanicalartworldwide.info.
The New Zealand exhibition opened 30 March 2018 at the Auckland Botanic Gardens. It will run until 1 July 2018. It will then travel to the Wellington Botanic Garden, to Blenheim’s Millennium Gallery, and possibly to Christchurch.
The two Wellington artists amongst the 36 participating New Zealand artists are:
Sue Wickison: Clianthus puniceus Kaka-beak (pen and ink); Dactylanthus taylorii The wooden rose (water colour);
Jane Humble: Passiflora tetrandra New Zealand passionfruit, ‘Kohia’ (water colour).
Wellington Botanical Society congratulates Sue and Jane on their outstanding skills in botanical art.
Baring Head / Orua pouanui
Baring Head, part of East Harbour Regional Park, contains the lower reaches of the Wainuomata River, along with about 60 ha of associated flood-plain and oxbows. Because it is a River of National Importance and contains, or is associated with, several rare and threatened habitats and species of flora and fauna, considerable eff ort is being committed to protecting and enhancing its ecological values.
Much of the river and most of the flood-plain have been fenced off from stock, woody weeds are being controlled, traps and bait-stations are targeting possums and predators, and we’ve planted about 12,000 seedlings so far. The benefits of this eff ort are already becoming evident, including natural recovery of the remaining native vegetation, and reduced bank slumping.
Our planting eff ort is now being accelerated through the Million Metres programme administered by the Sustainable Business Network. This involves crowdfunding, with details of our project being posted on their web site and the public having the opportunity to donate online. We’ve started very modestly this year to test the waters, so have a look at www.millionmetres.org.nz/openproject/2018-wainuiomata-restoration-at-baring-head.
Past work at Baring Head has shown that few species can cope with the combination of wind, dry summers and grass competition, and achieve rapid growth. The initial plantings will therefore focus on ngaio, cabbage trees and flax, which have proven to be hardy and vigorous on all parts of the site. These will be supplemented with toetoe in seeps, and rushes and Plagianthus divaricatus on the immediate river edge within the inundation zone.
Ngaio will be used across the terraces to create pockets of shade with reduced grass cover. We hope that there will be significant and rapid natural regeneration around these pockets, given the quantity of existing indigenous material within the grass. These pockets will break the wind, and create havens for lizards. They would also help the site survive a grass fire if that occurred. Ngaio provide rapid canopy cover and spreads to cover a wide area around each plant. We have already planted several strips and pockets across the site.
Cabbage trees and flax will be concentrated along the river banks to provide erosion control and shelter for other species that will be added (or self-introduce) later.
We are also taking the opportunity re-introduce forest species that are now largely absent from the lower river, by planting into areas that already have shelter. We have had permission to collect seedlings and seed from Rimutaka Forest Park, and will be focusing on the species present in the Catchpool Stream tributary of the river, and the one remaining significant remnant along the lower river. This will ultimately transform the river valley into a riverine forest.
Some rare and threatened species will be added into the river valley plantings as suitable sites become available. For example, we have already started adding new Muehlenbeckia astonii plants to supplement the two that are left, choosing our plants in accordance with recovery group advice. Plantings of inanga-friendly species such as Cyperus ustulatus will also help restore spawning sites.
In addition to this river-focused programme, we have a work-stream to improve vegetation on the coast and the escarpment above the river (the ‘grey scrub’ community).
Other initiatives are underway to increase the amount of community and corporate involvement in this project and the other planting programmes elsewhere at Baring Head. We’ll keep you posted on progress. As always, we would love to hear from you if you would like to help.
We now have volunteer accommodation at the lighthouse, and would welcome botanists who want to do more survey work in the area. Contact Paula Warren (pwarren (at) doc.govt.nz) for more information.
Colin Ryder, Treasurer, Friends of Baring Head
 |
Spiranthes novae-zelandiae. Photo: Jeremy Rolfe. |
Propagation of threatened orchid
Our research on the seed germination of the NZ ladies’ tresses (Spiranthes novae-zelandiae) has been published in the NZ Journal of Botany. In this article we described the steps we used to propagate this threatened orchid, and we also studied its genetic affinities with other Spiranthes species found in Australia and Asia.
After two years of inoculating the seeds, we have got plants that flowered and are still looking pretty healthy. Some have gone to Otari-Wilton’s Bush and others to Wellington Botanic Garden. This is the very first time a native orchid has been propagated from seed in NZ using its own mycorrhizal fungi. So we are over the moon! I would like to say a BIG THANKS to the Wellington Botanical Society for the funding provided. This has been super useful and key to our success!
Carlos Lehnebach (Jubilee Award) Jonathan Frerichs (Arnold Dench Award)
Our Land 2018 report - a call for action on environment
The Environmental Defence Society has commended the Ministry for the Environment and Stats New Zealand for their latest environmental report, Our Land 2018.
“This latest in the series of environmental reports under the Environmental Reporting Act 2015, focuses on the state of land and soil. It acknowledges some serious data gaps, but is otherwise a useful, insightful and frankly worrying analysis that shows mostly declining trends,” says EDS CEO Gary Taylor.
“In that respect it is similar to previous reports on other domains which have also shown mostly negative trends. We’ve got to do something and soon. “For example, it reveals that between 1996 and 2012 we lost 31,000 ha of tussock grassland – but we know that figure has dramatically increased since 2012. Similarly, it shows that large areas of indigenous shrubland, native forests and wetlands continue to diminish in spite of laws to protect them.
“Urban expansion is seeing productive and elite soils lost to development while exotic forest harvesting causes soil losses for 6–8 years. “Overall the report reveals that we are losing 192 million tonnes of soil a year, 44% of it from exotic grasslands. “Key drivers of loss, including of water quality, according to the report appear to be the 42% increase in the land area used by dairy farms in the period 2002–16 coupled with higher stocking rates.
“We know from previous reports about the extraordinarily high number of native species at risk from extinction on land. The only bright spot in that space is that 20 bird species are recovering because of active forest management. But that is an alarmingly small number against the total of about 215 bird taxa.
“What next? We can expect the Parliamentary Commissioner for the Environment to review the report and recommend responses. But mostly it reinforces the need for urgent action and provides irrefutable statistical support for the Government to push hard for big changes in the way we manage land.
“We consider it has a powerful and clear mandate from the electorate to do that.
“That means a suite of changes must be pushed forward including:
• a major overhaul of the Resource Management Act
• much better administration by regional councils
• deployment of a National Policy Statement on Indigenous Biodiversity
• strengthening the NPS Freshwater Management
• reviewing the National Environmental Standard on Plantation Forestry preparing a proper plan for the Billion trees
• investing more in pest and weed management
• more funding for DOC’s core functions
• reducing stocking rates especially in dairying
• better urban planning.
“It’s what this report leads to that’s important. It should be possible in 2018 to stop further decline in our natural environment and kick-start restoration of much of what’s been lost,” Mr Taylor concluded.
Source: Media Statement, Environmental Defence Society: Gary Taylor 021 895 896.
Weed control work in our region
 |
Cathedral bells (Cobaea scandens).
Photo: Jeremy Rolfe. |
What has been done so far this year in Kapiti, Porirua, Wellington and the Hutt Valley? Numerous environmental weeds (including banana passionfruit, blackberry, boneseed, buddleia, cherry, cathedral bells, climbing asparagus, Darwin’s barberry, Egeria, holly, hops, Japanese honeysuckle, Jerusalem cherry, gorse, hawthorn, iceplant, lupin, marram, old man’s beard, pines, Pinus contorta, selaginella, smilax, stinking iris, strawberry dogwood, sycamore, tradescantia, wild ginger, willow and yellow flag iris) have been targeted in our parks, reserves, Key Native Ecosystems (KNEs) and wetland areas etc. We have used several methods: removal by hand, cutting and stump treating, spraying herbicide from the ground, or aerially if there was no other practical option.
Where have we worked? Akatarawa Forest, Battle Hill Farm Forest Park, Baring Head KNE, Belmont Regional Park, Dry Creek, East Harbour Regional Park, Hayward Scenic Reserve, Haruatai Swamp Forest KNE, Hutt Water Collection Area, Kaitoke Regional Park, Karehana Bay KNE, Keith George Memorial Park, Korokoro Reserve, Lower Waikanae Bush Fragments KNE, Nga Manu Wetland Complex KNE, O te Pua Pukehou Wetland KNE, Otaki Coast KNE, Pakuratahi Forest, Parangarahu Lakes, Peka Peka Dunes KNE, Speedys Reserve, Waikanae River KNE, Pauatahanui, and Porirua Stream Mouth & Estuary Enhancement site, Pukerua Coast KNE, Trentham Memorial Park, Te Hapua Wetland Complex KNE, Waterfall Bush Road KNE, Whitireia Park, and several small private wetland programme areas.
Why are we doing this work? Some sites are in the initial stages of control where much work is needed to bring invasive species under control. The level of control needed reduces significantly over the years, and at varying intervals of time, we arrive at a situation where the site is relatively free of threats and just needs regular maintenance to nip threats in the bud.
Regional Pest Management Plan species
 |
Jerusalem cherry (Solanum pseudocapsicum).
Photo: Jeremy Rolfe. |
We have monitored and controlled all known moth plant, woolly nightshade, perennial nettle, senegal tea, purple loosestrife and manchurian wild rice sites; as well as surveyed to determine the extent of newly discovered sites, e.g., moth plant near Waterloo Station.
We have had many discussions about the species to be included in our upcoming new RPMP, and what the rules would be for these species.
We’ve dealt with 26 boundary complaints – these are generally about blackberry, gorse or old man’s beard. We also get numerous general enquiries to respond to.
Biocontrol
This year we have monitored green thistle beetles, tradescantia beetles, and buddleia. We have released tradescantia beetles, Japanese honeysuckle butterfly, broom gall mites, Darwin’s barberry weevils, and tradescantia rust – for which we have high hopes of it making a difference.
NPPA
104 nursery or plant outlets have been inspected so far this financial year. No unwanted organisms were found at any of these.
Check Clean Dry
Our Check Clean Dry programme finished for the year in March. The message was spread through waterside advocacy, presentations at club meetings, school visits, social media and the GWRC web site.
If you would like more information on particular species or areas, please contact me.
Katrina Merrifield, Biosecurity Officer (Plants), Greater Wellington Regional Council, Te Pane Matua Taiao
Go to
Earlier News 2016-17.

 Site Index
Site Index







 Site Index
Site Index





